2016 Archived Content
November 3-4, 2016

11th Annual
Aligning Portfolio and Productivity with Corporate Strategy to Drive Innovation
In an environment of declining revenues and uncertain commercial success, biopharma, device and R&D companies need to continuously evaluate their portfolios. Manufacturers must make tough decisions about which products and projects to pursue to optimize long-term revenue and reduce overall risk. Cambridge Healthtech Institute’s and the BioPharma Strategy Series’ 11th Annual “Portfolio Management: Aligning Portfolio and Productivity with Corporate Strategy to Drive Innovation” attracts over 100 senior R&D executives from the pharma, biotech, device, IT, public and governmental communities who share best practices in project and portfolio management, R&D innovation, decision analysis, forecasting and the improvement of operational models. Effective portfolio management requires the alignment of portfolio and productivity with corporate strategy, supporting agile development and adaptation of data into planning.
Thursday-Friday, November 3-4, 2016
Thursday, November 3
Session Break, Transition to Special Welcome Luncheon
This year, the joint keynote session will begin with a networking Welcome Luncheon and Presentation. Attendees from Strategic Resource Management and Portfolio Management are invited to enjoy a delicious plated luncheon while networking and collaborating with fellow attendees during an informative “Lunch-and-Learn” presentation.
12:15 pm WELCOME LUNCHEON PRESENTATION: Embracing Real-Time Portfolio Management to Build Trust and Make Effective Decisions
 Richard Sonnenblick, Ph.D., CEO, Enrich Consulting
Richard Sonnenblick, Ph.D., CEO, Enrich Consulting
Portfolio management is difficult because of its myriad moving parts: Multiple decision makers, tens of projects, dozens of project team members, hundreds of inputs, and thousands of changes to project details leading up to a portfolio review. In this talk, we’ll discuss what it takes to create project-level scenarios, keep audit trails of all project changes, and report on every aspect of a portfolio in real-time, without relying on labyrinthine spreadsheets or endless PowerPoint decks.
1:25 Welcome Portfolio Management Conference Registrants & Joint Session Announcements
 Micah Lieberman, Executive Director, Conferences, BioPharmaceutical Strategy Series, Cambridge Healthtech Institute (CHI)
Micah Lieberman, Executive Director, Conferences, BioPharmaceutical Strategy Series, Cambridge Healthtech Institute (CHI)
1:30 Chairperson’s Opening Remarks for Shared Keynote Session
 Charles Alsdorf, Director and Capital Efficiency Leader, Deloitte Transactions and Business Analytics LLP
Charles Alsdorf, Director and Capital Efficiency Leader, Deloitte Transactions and Business Analytics LLP
1:35 Leveraging the Wisdom of the Crowd to Optimize Your Portfolio
 Robert Boland, Associate Director, Emerging Science and Innovation Strategy, Johnson & Johnson
Robert Boland, Associate Director, Emerging Science and Innovation Strategy, Johnson & Johnson
Prediction markets and crowdsourcing have been leveraged for some time by some of the biggest companies in the world. Discover how companies such as Johnson & Johnson are leveraging these innovative approaches in unique ways to capitalize on the knowledge of their employees, while driving strategic insights and key decision making on some of their most important portfolios.
2:15 Strategic Resource Management and the Value of an Enterprise System to Enhance R&D Productivity and Inform Business Decisions
 Jean Lee, Ph.D., Vice President, Portfolio & Project Management, Global Product Development, Pfizer R&D
Jean Lee, Ph.D., Vice President, Portfolio & Project Management, Global Product Development, Pfizer R&D
In a constantly evolving business environment, R&D leaders are utilizing the totality of the information to inform project decisions and optimize their portfolios. A number of case studies will be shared to demonstrate how a fully integrated planning and forecasting system, which includes all projects in the portfolio from candidate nomination to LOE, has been utilized to drive business decisions. The utility of the system for scenario planning and to support alliances/co-development projects will be discussed.
2:45 Diversification Strategies in Pharmaceutical Portfolios: Hedging Downside and Upside Risk?
 Jonathan Freeman, Ph.D., Senior Vice President and Global Head, Strategy Development & Portfolio Management, EMD Serono
Jonathan Freeman, Ph.D., Senior Vice President and Global Head, Strategy Development & Portfolio Management, EMD Serono
Through lifecycle management and other lower-risk diversification strategies, pharmaceutical companies have become familiar if not adept at covering downside "catastrophic" risk scenarios. Although these approaches ensure comfort in delivering market-tracking growth, by definition they cannot be expected to offer outperform potential. How can pharma ensure that it can retain upside risk in its portfolio at a price that it can afford, and can that price be reduced by turning the unilateral risk into collective uncertainty through effective risk pooling and partnering?
3:15 INTERACTIVE PANEL DISCUSSION: Opportunities and Leading Practices in R&D-Commercial Collaboration
Historically, the R&D and Commercial functions in biopharma companies have operated fairly independently from each other, passing the proverbial baton along the product lifecycle. As a result, some products fail to come to market or, when they do, they may not meet their true potential. In this panel discussion, we will hear about the opportunities for earlier and deeper collaboration, as well as examples where this collaboration has made a difference:
- What has been the collaboration environment and what factors have contributed to these functions operating independently and sequentially?
- What are some recent examples where greater collaboration is leading to tangible results?
- What does that collaboration look like?
- What can everyone learn from the medical devices field where the R&D and engineering functions are very close to the marketplace, including customers, payers and patients?
Moderator:
 David Davidovic, Founder, pathForward; former VP and Global Head, Commercial Services, Genentech and Roche
David Davidovic, Founder, pathForward; former VP and Global Head, Commercial Services, Genentech and Roche
Panelists:
 Richard Murray, M.D., Vice President and Deputy Chief Patient Officer, Merck
Richard Murray, M.D., Vice President and Deputy Chief Patient Officer, Merck
 Stephanie Brown, MBA, Vice President, Head Specialty Business Unit, Takeda Pharmaceuticals US
Stephanie Brown, MBA, Vice President, Head Specialty Business Unit, Takeda Pharmaceuticals US
 Svetlana Pidasheva, Ph.D., Director, Global Scientific Communications and Publications, Sanofi
Svetlana Pidasheva, Ph.D., Director, Global Scientific Communications and Publications, Sanofi
4:00 Breakout Discussion Groups Introduction
4:05 Refreshment Break with Exhibit Viewing
Interactive Breakout Discussions
Concurrent breakout discussion groups are interactive, guided discussions hosted by a facilitator or set of co-facilitators to discuss some of the key issues presented earlier in the day’s sessions. Delegates will join a table of interest and become an active part of the discussion at hand. To get the most out of this interactive session and format please come prepared to share examples from your work, vet some ideas with your peers, be a part of group interrogation and problem solving, and, most importantly, participate in active idea sharing.
The Breakout Discussion session will be run two times, on Wednesday afternoon just before the reception and Thursday afternoon at the end of the day.
TABLE 1: Using Project Milestones as Principle Objectives of Development (PODS) for Resource Planning and Portfolio Management
Moderators:
 Cleat Jerden, Director Strategic Planning and Operations, CMC Integration, Amgen
Cleat Jerden, Director Strategic Planning and Operations, CMC Integration, Amgen
 Samantha Fairbairn, Senior Manager Strategic Planning and Operations, Attribute Sciences, Amgen
Samantha Fairbairn, Senior Manager Strategic Planning and Operations, Attribute Sciences, Amgen
- How do development functions align on key milestones and activities to plan their resources and work?
- Are portfolio management software programs improving portfolio planning?
- Are capacity management and portfolio management managed through the same team, or are they separate teams with different outputs?
TABLE 2: Partner vs. Vendor: Are You Getting the Value Out of Your Vendor Management Strategy?
Moderators:
 Matthew Kokkonen, Associate Director, IT, BioMarin Pharmaceutical LLC
Matthew Kokkonen, Associate Director, IT, BioMarin Pharmaceutical LLC
 Larry Radowski, MBA, Managing Director, Strategy and Business Development, Integrated Project Management, Inc. (IPM)
Larry Radowski, MBA, Managing Director, Strategy and Business Development, Integrated Project Management, Inc. (IPM)
 Tom Defay, Ph.D., Defay Drug Discovery, Program Management, and Informatics, LLC
Tom Defay, Ph.D., Defay Drug Discovery, Program Management, and Informatics, LLC
- Each year services firms receive ~$2 Billion for impractical data and poorly implemented recommendations (per HBR). To minimize this risk, clients should better understand what consulting and consultants can actually accomplish: Likewise, consultancies need to better understand and deliver per industry’s needs today.
- What does industry need from a consultancy? How have you benefited from a consultancy? (e.g. Information vs. Problem Solving, Referee vs. Consensus Building, Outsourcer vs. Life-long Adviser)
- What does a consultancy need from industry in order to succeed? (e.g. frameworks vs. work accomplished)
- How effective are “best practices” when managing consulting resources around vendor management/consultancy at meeting organization’s needs today? Is a company’s structure and culture aligned with how they partner with consultancies?
TABLE 3: Quantification of Uncertainty: What Can We and What Should We Quantify?
Moderators:
 Daniel Zweidler, Ph.D., Senior Fellow, Mack Institute for Innovation Management, The Wharton School, UPenn; President, DZA, Inc.
Daniel Zweidler, Ph.D., Senior Fellow, Mack Institute for Innovation Management, The Wharton School, UPenn; President, DZA, Inc.
 Robert Boland, MBA, Associate Director, Emerging Science and Innovation Strategy, Johnson & Johnson
Robert Boland, MBA, Associate Director, Emerging Science and Innovation Strategy, Johnson & Johnson
- Historical perspective, how good are we at predicting revenues (please come prepared with real examples)
- When do you know that you have an impactful drug in your pipeline?
- How does this impact your decision making?
TABLE 4: Portfolio Management at The Speed of Light: The Impact of Game Changing Therapies On Portfolio Planning
Moderators:
 Jeff Mumford, Managing Director, East Region, Integrated Project Management, Inc. (IPM)
Jeff Mumford, Managing Director, East Region, Integrated Project Management, Inc. (IPM)
 John Phillips, Managing Director, Life Science Lead for Capital Efficiency Practice, Deloitte Transactions and Business Analytics LLP
John Phillips, Managing Director, Life Science Lead for Capital Efficiency Practice, Deloitte Transactions and Business Analytics LLP
 Richard Sonnenblick, Ph.D., CEO, Enrich Consulting
Richard Sonnenblick, Ph.D., CEO, Enrich Consulting
- What is the impact that the new immuno-oncology wave has had on regulatory time lines, strategies and ability to plan portfolios?
- What Therapeutic Areas are showing the greatest opportunity and also bringing new challenges?
TABLE 5: Optimizing Resource Management’s and Your PMO Office’s Business Impact: Data vs. Emotions in Decision Making?
Moderators:
 Richard Wilner, Director, North America, Portfolio Management Office, Sanofi
Richard Wilner, Director, North America, Portfolio Management Office, Sanofi
 Charles Dormer, Principal, APEX STP
Charles Dormer, Principal, APEX STP
 Jared Fantasia, Head, Global Portfolio & Project Management Operations (GPPM), Johnson & Johnson
Jared Fantasia, Head, Global Portfolio & Project Management Operations (GPPM), Johnson & Johnson
- What is the business case for resource management? What impact do you expect it to have on your business?
- How can you create a culture from being driven by anecdotes and opinions, to being driven by insights based on data? Have you done this?
- How can an organization like a PMO change the organization through influence without requiring or relying on solid-line reporting authority?
- Are your decision driven by data or emotion?
TABLE 6: Consolidation and Break-Ups Among Life Sciences Companies and Vendors: What are the Trends and What do They Mean?
Moderators:
 David Davidovic, Founder, pathForward; former VP and Global Head, Commercial Services, Genentech and Roche
David Davidovic, Founder, pathForward; former VP and Global Head, Commercial Services, Genentech and Roche
 John Fischer, MBA, Project Management Consultant, Integrated Project Management Company, Inc. (IPM)
John Fischer, MBA, Project Management Consultant, Integrated Project Management Company, Inc. (IPM)
 Charles Alsdorf, Managing Director and Capital Efficiency National Practice Leader, Deloitte Transactions and Business Analytics LLP
Charles Alsdorf, Managing Director and Capital Efficiency National Practice Leader, Deloitte Transactions and Business Analytics LLP
- Life sciences companies have been consolidating with M&A activity for years. Will this trend continue or will some behemoths break up into pieces?
- Vendors into the industry have been consolidating in efforts to expand product and service portfolios and further penetrating their customer bases. Will this continue? Who wins in these mergers?
- How are companies managing transitions to minimize disruption?
TABLE 7: Leveraging Private-Public Partnerships and Alternative Business Models to Drive Innovation in Global Health
Moderators:
 Thomas Forissier, Deputy Director, Strategy Planning & Management, Global Health, Bill & Melinda Gates Foundation
Thomas Forissier, Deputy Director, Strategy Planning & Management, Global Health, Bill & Melinda Gates Foundation
 George Santangelo, Ph.D., Director, Office of Portfolio Analysis (OPA), Division of Program Coordination, Planning, and Strategic Initiatives (DPCPSI),National Institute of Health (NIH)
George Santangelo, Ph.D., Director, Office of Portfolio Analysis (OPA), Division of Program Coordination, Planning, and Strategic Initiatives (DPCPSI),National Institute of Health (NIH)
- How can both the Public and the Private partners achieve a sustainable model for both parties?
- What would it take for the private sector to commit best resources and assets to global health (beyond corporate social responsibility)? What incentives need to be in place? What might be the levers to lower the cost of opportunity? Where might the first areas of collaboration be (i.e., where should we start)?
- What have we learned from past examples on how to structure such endeavors?
View Breakout Discussion Groups Page
Close of Strategic Resource Management Executive Forum (Portfolio Management continues on through Friday, November 4)
Friday, November 4
7:45 am Morning Coffee
8:15 Chairperson’s Remarks
 Robert Boland, Associate Director, Emerging Science and Innovation Strategy, Johnson & Johnson
Robert Boland, Associate Director, Emerging Science and Innovation Strategy, Johnson & Johnson
8:20 FEATURED PRESENTATION: Centralized vs. Decentralized Portfolio Management
 Ian Popoff, Senior Director, Strategy and Portfolio Management, Pfizer
Ian Popoff, Senior Director, Strategy and Portfolio Management, Pfizer
Co-developed by Gautam Gupta, MBA, Vice President, Strategy, Portfolio, Competitive Intelligence and Enterprise Operations, Worldwide Research and Development, Pfizer
This presentation will focus on how to optimize the balance between centralized (i.e., corporate) and decentralized (i.e., TAs/Line Functions) control of portfolio management processes like portfolio prioritization, allocation of funding, go/no-go decisions, etc. There are pros and cons of either approach – centralized processes tend to bring more consistency across TAs/lines and allow one to optimize across the entire portfolio whereas decentralized processes tend to be faster/more efficient, they empower the lines and also leave decision making to the subject matter experts. So, what is the best approach?
9:00 A Holistic Approach to Strategic Resource & Portfolio Management
 Ken Dobie, Ph.D., MBA, Founder, Skyemar Consulting LLC; former Director, Corporate Planning & Portfolio Management, Illumina
Ken Dobie, Ph.D., MBA, Founder, Skyemar Consulting LLC; former Director, Corporate Planning & Portfolio Management, Illumina
Our Corporate Planning & Portfolio Management function supports a holistic approach to aligning corporate strategies and priorities with the project portfolio and associated resources. First, we support the annual strategic planning process in collaboration with senior management to produce our three year product development roadmaps and Strategic Plan articulating our strategic top down guidance. Second, we support the cascade of strategic priorities into tactical goals represented as Operating Plans for the coming year. Third, we lead project portfolio and resource management activities spanning Research, Product Development, Operations, and individual Business Units. This supports the roll-up of the portfolio status at quarterly reviews where portfolio investment/prioritization decisions are made.
9:30 Managing Shared Resources: How to Optimally Assess, Acquire, and Allocate In-Demand Resources across the Competitive Landscape of Your Portfolio
 John Fischer, MBA, Project Management Consultant, Integrated Project Management Company, Inc. (IPM)
John Fischer, MBA, Project Management Consultant, Integrated Project Management Company, Inc. (IPM)
Life Sciences companies are very commonly challenged to deliver large portfolios of projects using shared resources. This problem is complex as internal competition can be significant in pursuit of resources with specific talents and expertise, and who are typically assigned to multiple projects. Without the right structure, processes, and discipline in managing the portfolio of projects and the resource pool, shared resources can become overloaded and unavailable to support the organization’s greatest priorities, thereby negatively impacting the organization’s collective ability to deliver. This session will focus on strategies and tactics to improve managing shared resources across the portfolio and improve organizational portfolio performance.
10:00 Coffee Break with Exhibit Viewing
10:45 CASE STUDY: Process and Methodology to Evaluate the Decision to Progress Internal and/or External Assets
 Elayne Ko, Director, Portfolio and Decision Analysis, Pfizer
Elayne Ko, Director, Portfolio and Decision Analysis, Pfizer
When opportunities exist to progress an internal pharma R&D compound or to pursue in-licensing of an external compound, a decision on the path forward for one and/or both compounds is needed, and typically a recommendation is required within a short timeframe. This case study describes the process of establishing separate evaluation teams and developing methodologies to support the decision analyses within the broader portfolio context. The presentation highlights issues with evaluating internal and external programs in parallel, potential areas of bias along the decision-analytic process, and areas of focus during the facilitation process in reaching alignment on the recommendation.
11:15 CO-PRESENTATION & INTERACTIVE DISCUSSION: Incorporating Strategic Risk into Portfolio Decisions and Valuations
 Charles Alsdorf, Managing Director and Capital Efficiency National Practice Leader, Deloitte Transactions and Business Analytics LLP
Charles Alsdorf, Managing Director and Capital Efficiency National Practice Leader, Deloitte Transactions and Business Analytics LLP
 John Phillips, Managing Director, Life Science Lead for Capital Efficiency Practice, Deloitte Transactions and Business Analytics LLP
John Phillips, Managing Director, Life Science Lead for Capital Efficiency Practice, Deloitte Transactions and Business Analytics LLP
The life science industry is constantly undergoing significant transformation due to scientific innovation, M&A, economics, regulatory and legislative changes, new and emerging technology and customer preference changes to name just a few issues. Your organization is dealing with a competitive landscape which is transforming, refreshing your strategy, and testing underlying assumptions. The demands on your portfolio, organic and/or externally sourced, to generate significant returns are increasing, and your organization may lack the portfolio processes, systems, and capabilities to identify and respond to strategic risks. This interactive session will focus on identifying how you identify and address those risks beyond just PTRS that have a material impact on the long-term performance of your portfolio. During the session, we will exchange views and examine the audience’s experiences in:
- spotting trends and disruptors
- testing assumptions underlying portfolio decisions
- identifying and providing insights on their strategic risks
- how those risks impact portfolio management
 11:45 Co-Luncheon Presentation: How R&D Operational Excellence can Contribute to Superior Portfolio Management Outcomes
11:45 Co-Luncheon Presentation: How R&D Operational Excellence can Contribute to Superior Portfolio Management Outcomes
 Nicole Cohen, Managing Director, Life Sciences R&D Consulting Practice Lead, Accenture
Nicole Cohen, Managing Director, Life Sciences R&D Consulting Practice Lead, Accenture
 Marc Philipp, Senior Manager, Life Sciences R&D Strategy, Accenture
Marc Philipp, Senior Manager, Life Sciences R&D Strategy, Accenture
To maximize business value from R&D portfolio management, pharma companies need to be fast in taking the right decisions in a highly volatile and uncertain environment. Despite a steady advancement of applied methodologies, metrics and tools, many players - especially Big Pharma - still underestimate the positive impact of R&D operational excellence initiatives. This presentation will discuss three key levers of R&D operational excellence and outline their contribution to superior portfolio management outcomes.
1:25 Chairperson’s Remarks
 Larry Radowski, Managing Director, Strategy and Business Development, Integrated Project Management
Larry Radowski, Managing Director, Strategy and Business Development, Integrated Project Management
1:30 Predictive Analytics and Economic Valuation in Portfolio Management of Life Sciences Assets
 Gill Eapen, Managing Director, Business Advisory, Stout Risius Ross (SRR)
Gill Eapen, Managing Director, Business Advisory, Stout Risius Ross (SRR)
Life sciences assets show high uncertainty in investments needed, timelines, technical risks and market benefits. Predictive analytics could be applied to define these parameters and then fed into an economic valuation framework to assess the value and risk to allow better selection, design and resourcing decisions.
2:00 Looking Before You Leap: A Case Study to Build a Value Focused Project Portfolio Management Office
 Matthew Kokkonen, Associate Director, IT, BioMarin Pharmaceutical LLC
Matthew Kokkonen, Associate Director, IT, BioMarin Pharmaceutical LLC
Companies often struggle to build portfolio and project management offices that meet the diverse stakeholder interests that they serve. Building a portfolio or project management office often pits proponents of ‘best practice’ against the ultimate mission of a PMO, which is to optimize the portfolio’s performance and ensure maximal value delivery from the programs and projects under its control. This presentation will explore to how to implement ‘best practice’ and build the foundation for a pragmatic PMO using a flexible portfolio management tool.
2:30 CO-PRESENTATION AND INTERACTIVE SESSION: The Importance of Supportive Business Processes in Portfolio Management
Part 1: A Fool with a Tool is Still a Fool
 Mark Lane, Ph.D., Consultant, TayganPoint Consulting Group
Mark Lane, Ph.D., Consultant, TayganPoint Consulting Group
Part 2: Optimizing Portfolio Decision-making
 Sushil Rijhwani, Ph.D, MBA, PMP, Senior Director, Project & Portfolio Management, Global Project Management & Leadership, Daiichi-Sankyo
Sushil Rijhwani, Ph.D, MBA, PMP, Senior Director, Project & Portfolio Management, Global Project Management & Leadership, Daiichi-Sankyo
Part 3: Real Life Challenges & Success Stories When Creating a Single Source of Truth (SSOT)
 Jared Fantasia, Head, Global Portfolio & Project Management Operations (GPPM), Johnson & Johnson
Jared Fantasia, Head, Global Portfolio & Project Management Operations (GPPM), Johnson & Johnson
Despite employing the most up to date and elegant software tools and analytics most portfolio management approaches are not delivering maximal business benefit. Although there may be many contributing factors the main drivers for this include an inability to establish a single, validated data repository (the elusive single source of truth), failing to construct and conduct a portfolio governance process focused on end to end value creation and fact-based decision-making and ineffective change management when implementing new business processes and software. This session will explore these foundational aspects needed to drive optimal portfolio management practices.
3:25 Chairperson’s Closing Remarks
3:30 Close of Portfolio Management Executive Forum
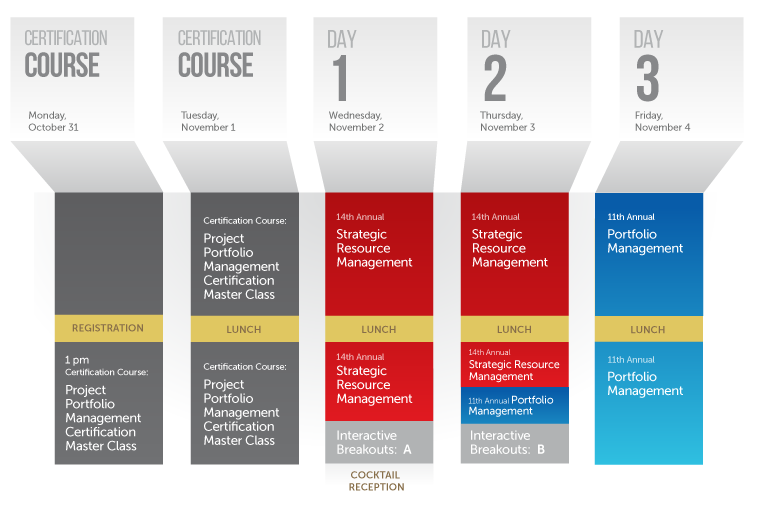
Again this year, we will be running the meeting back to back with the 14th annual meeting focused on Strategic Resource Management, which takes place November 2-3, 2016, creating a three-day event (Nov. 2-4) covering the topics most important to you. There is also a Monday-Tuesday Project Portfolio Management Certification Master Class this year, which gives attendees a five-day event option (see map below).
Group Discounts and NIH/Government Discounts Available! Special rates are available for multiple attendees from the same organization and for NIH/Government employees. For more information on group and government discounts contact Cheryle Rosenberg at 781-972-5489
For exhibit and partnering information, please contact:
Uma Patel
Business Development Manager
Cambridge Healthtech Institute (CHI)
T: (+1) 781.972.1349
E: upatel@healthtech.com